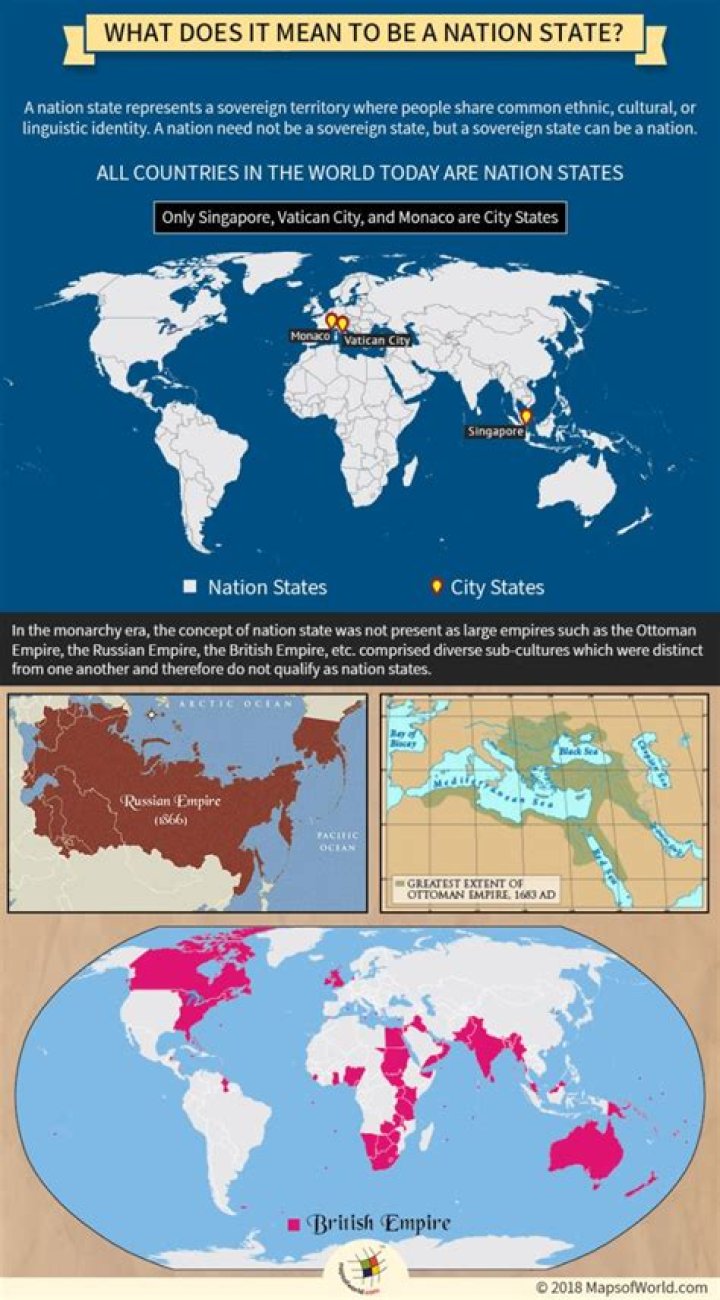
Why is KMnO4 purple in colour

Charlotte Adams
Published May 06, 2026
The purple colour of KMnO4. is due to the charge transfer transition. Reason- The intense colour is most of the transition metal complexes is due to d-d transition.
What gives potassium permanganate its color?
The deep purple color of potassium permanganate is caused by the movement of an electron to a vacant d-orbital in the manganese atom. The transition occurs when the chemical is in the presence of light. This is confirmed by the empty 3d-orbital in manganese.
What ion makes potassium permanganate purple?
Permanganate salts may also be generated by treating a solution of Mn2+ ions with strong oxidants such as lead dioxide (PbO2), sodium bismuthate (NaBiO3), or peroxydisulfate. Tests for the presence of manganese exploit the vivid violet color of permanganate produced by these reagents.
Why is the solution purple in colour?
Pure iodine is violet, but when it’s dissolved in water, it accepts an electron from the oxygen atom, affecting how it absorbs light. When you shake the fluids, the iodine leaves the water and dissolves in the oil, and returns to its purple colour!What colour is potassium permanganate?
The potassium permanganate solution should be a pink colour.
Why is potassium Manganate colorless?
The solution will gradually become colourless as the sulfur dioxide reacts with the potassium permanganate. The colour of the manganate ion is lost as the sulfurous acid is oxidized to sulfuric acid.
Why is KMnO4 yellow?
The observed colour may be caused by a cloud of finely dispersed particles of dark brown manganese(IV) oxide (MnO2), which dissolves when the solution is well mixed again so that the intended reaction continues.
How do you acidify potassium permanganate?
Add 250 mL of purified water (freshly boiled and cooled) and 10 mL sulfuric acid (96% H2SO4, sp g 1.84). Add rapidly from a buret about 95% of the theoretical quantity of potassium permanganate solution needed; stir until the solution is clear.Why does water become Coloured when crystals of potassium permanganate is added?
Answer: Because of the collision of water molecules. Explanation: When potassium permanganate crystals are introduced in the water, the water molecules collide with the surface of the crystals and thus the water becomes coloured.
What is the colour of potassium permanganate in water?Potassium permanganate (KMnO4) is an odorless solid, and they look like crystals of dark purple or bronze color. When these crystals are dissolved in water, the solution becomes purple in color.
Article first time published onWhen potassium permanganate dissolve in water the color of the solution is?
It is a purplish-black crystalline solid, that dissolves in water to give intensely pink or purple solutions. Potassium permanganate is widely used in chemical industry and laboratories as a strong oxidizing agent, and also as a medication for dermatitis, for cleaning wounds, and general disinfection.
Is iodine purple or brown?
Iodine is violet when dissolved in carbon tetrachloride and saturated hydrocarbons but deep brown in alcohols and amines, solvents that form charge-transfer adducts.
Why does potassium Manganate turn from purple to Colourless?
When we bubble in the sulphur dioxide gas into the purple acidified potassium permanganate solution the manganate ion is reduced by sulphur dioxide as seen in the reaction below. … Hence, $S{O_2}$ turns acidified potassium permanganate from purple to colourless.
What is the colour of permanganate ion?
The ion has tetrahedral geometry. Permanganate solutions are purple in color and are stable in neutral or slightly alkaline media.
What solution changes acidified potassium manganate VII solution from purple to Colourless?
If the potassium manganate(VII) solution is acidified with dilute sulphuric acid, the purple solution becomes colourless.
What is the color of potassium?
Potassium metal is soft and white with a silvery lustre, has a low melting point, and is a good conductor of heat and electricity. Potassium imparts a lavender colour to a flame, and its vapour is green. It is the seventh most abundant element in Earth’s crust, constituting 2.6 percent of its mass.
Why does the colour of potassium permanganate not disappear when?
Explanation: Initially colour disappears because colouredpermanganate ions of potassium permanganate are consumed to oxidiseethanol. When excess is added colour does not change because there is nomore alcohol left and hence there is noreaction.
What color is absorbed by a permanganate solution?
Potassium permanganate solution has such high deep purple/ violet color because it absorbs the green or the green-yellow color between 500-550 nm as shown in Figure 1.
Why do redox reactions cause color change?
The reactants have converted to products, and since reactants and products may be of a different colour, you therefore observe a change in colour. This is likely due to a change in oxidation state of the metal central atom of the complex you are reacting.
What is colour of potassium dichromate?
Potassium dichromate (K2Cr2O7) The crystals of potassium dichromate are orange or orange red in colour, and it was first used in biological studies by MÜller in 1859.
What is the colour of potassium iodide?
Potassium iodide is a white crystalline salt with chemical formula KI, used in photography and radiation treatment.
Is permanganate an oxidizing agent?
Potassium permanganate, KMnO4, is a powerful oxidizing agent, and has many uses in organic chemistry.
Why is potassium iodide Brown?
Potassium Iodide (KI) can be very useful because when it is oxidised a colour change is produced. The Iodide ion (I-) is oxidised to iodine(I2). The colour of the solution will change from colourless to yellow-brown. If you then add starch indicator it will turn blue-black.
Why is potassium permanganate acidified?
Potassium manganate(VII) (potassium permanganate) is a powerful oxidising agent. Potassium manganate(VII) is usually used in neutral or alkaline solution in organic chemistry. Acidified potassium manganate(VII) tends to be a rather destructively strong oxidising agent, breaking carbon-carbon bonds.
When a crystal of potassium permanganate is placed in a beaker containing water is purple Colour spread throughout the water?
What does this observation tell us about the nature of potassium permanganate and water? Solution: The observation of purple colour spreads throughout the water is that the crystal of potassium permanganate is made up of many small particles and these particles of water have spaces between them. 28.
Why did the colour move through the water in diffusion?
The food coloring mixes through the hot water faster than it mixes with the cold water. This is because in hot water, the water molecules have more energy and are moving faster than the molecules of cold water.
What happens when potassium permanganate is added to water?
Potassium permanganate oxidizes iron, manganese, and hydrogen sulfide into particles. … If too much potassium permanganate is fed into the water prior to filtration, the excess potassium permanganate serves as a regenerant for the filter media. The water should be colorless when it leaves the filter.
Can I drink potassium permanganate?
BACKGROUND: Potassium permanganate is used clinically as an antiseptic and antifungal agent. Ingestion of potassium permanganate may result in damage to the upper gastrointestinal tract. Burns and ulceration of the mouth, esophagus and stomach occur due to its action.
How do you test for oxidising agent?
- Test for Oxidising Agent: Use of a Reducing Agent e.g. aqueous potassium iodide, KI(aq) Observation: Colourless solution turns brown. …
- Test for Reducing Agent: Use of an Oxidising Agent e.g. acidified potassium manganate (VII) …
- YouTube Video Tutorial. Let’s take a look at the YouTube Video now….
What reacts with potassium dichromate?
Reacting the sample with an excess of potassium dichromate, all ethanol is oxidized to acetic acid: CH3CH2OH + 2[O] → CH3COOH + H2O. Full reaction of converting ethanol to acetic acid: 3 C2H5OH + 2 K2Cr2O7 + 8 H2SO4 → 3 CH3COOH + 2 Cr2(SO4)3 + 2 K2SO4 + 11 H2O.
How do you remove potassium permanganate stains?
- Scrub the skin with lukewarm water if the stain is on the hands or other exposed part of the body. …
- Treat the stain with a diluted hydrochloric acid. …
- Rinse stained papers with sulfurous acid. …
- Use sodium metabisulfite to remove potassium permanganate stains from all surfaces.