What type of molecule is CBr4

Dylan Hughes
Published Apr 21, 2026
Name of MoleculeCarbon tetrabromideNatureNonpolarTotal Valence electron for CBr432
What is CBr4 used for?
Uses. It is used as a solvent for greases, waxes, and oils, in plastic and rubber industry for blowing and vulcanization, further for polymerization, as a sedative and as an intermediate in manufacturing agrochemicals. Due to its non-flammability it is used as an ingredient in fire-resistant chemicals.
What does CBr4 look like?
Carbon tetrabromide appears as a colorless crystalline solid. Much more dense than water and insoluble in water.
What kind of bond is CBr4?
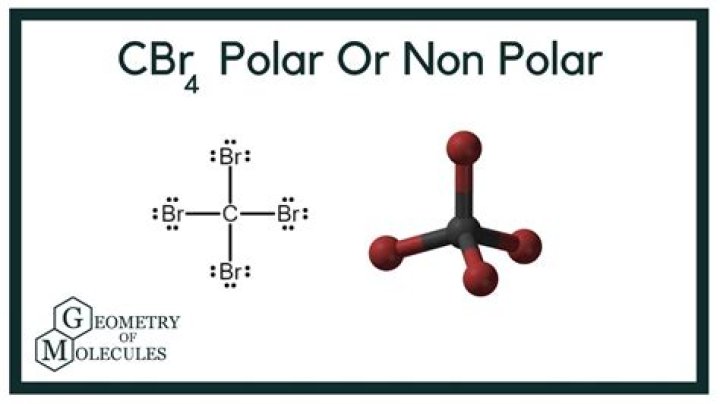
The bond formed in carbon tetrabromide is covalent since the constituent atoms, carbon and bromine, are non-metals. CBr4 C B r 4 has tetrahedral structure, in which 4 bromine atoms are substituted symmetrically around the central carbon atom and hence the dipole moment gets cancelled makes the compound non-polar.What is the empirical formula for CBr4?
Chemical FormulaCBr4IUPAC NametetrabromomethaneSMILES StringBrC(Br)(Br)BrInChIInChI=1S/CBr4/c2-1(3,4)5InChIKeyHJUGFYREWKUQJT-UHFFFAOYSA-N
What is the correct IUPAC name for ClF3?
PubChem CID24637StructureFind Similar StructuresChemical SafetyLaboratory Chemical Safety Summary (LCSS) DatasheetMolecular FormulaClF3SynonymsCHLORINE TRIFLUORIDE Chlorotrifluoride 7790-91-2 Chlorine fluoride (ClF3) Trifluorure de chlore More…
Is CBr4 polar or non polar?
The CBr4 molecule is non-polar. … Both CBr4 and CH3Br have four regions of electrons around the central carbon atom. These are all bonding electron regions (clouds) so the shape of both molecules is tetrahedral. The C-Br bond is polar due to the difference in electronegativity between C and Br.
What is the total number of electron groups in CBr4?
The Lewis structure for CBr4 C B r 4 is, (a.) There are 12 electron groups (3 electron groups on each Br atom).What is the correct name for N2O3?
PubChem CID61526Chemical SafetyLaboratory Chemical Safety Summary (LCSS) DatasheetMolecular FormulaN2O3SynonymsDinitrogen trioxide NITROGEN TRIOXIDE Nitrogen oxide (N2O3) N-oxonitramide 10544-73-7 More…Molecular Weight76.012
How many bonds are present in the molecule CBr4?The Carbon tetrabromide molecule contains a total of 4 bond(s) There are 4 non-H bond(s). The 2D chemical structure image of Carbon tetrabromide is also called skeletal formula, which is the standard notation for organic molecules.
Article first time published onIs mgf2 ionic or covalent?
Magnesium fluoride is a binary ionic compound. It is comprised of two fluorine and one magnesium atoms.
What is the name of HCN AQ?
Name of AnionName of AcidExamplesHypo…- iteHypo…- ous acidHClO hypochlorite hypochlorous acid
How do you name N2S4?
N2S4 is a binary covalent compound, not an ionic compound and it’s name is dinitrogen tetrasulfide. Nitrogen and sulfur do not form an ionic compound. The electronegativity difference between them is 0.48, which makes the N-S bond barely polar covalent. Ionic bonds have an electronegativity difference greater than 1.6.
What is the proper name for n2o5?
PubChem CID66242Molecular FormulaN2O5SynonymsDinitrogen pentaoxide Dinitrogen pentoxide Nitrogen pentoxide 10102-03-1 nitro nitrate More…Molecular Weight108.01DatesModify 2021-12-18 Create 2005-03-27
What is the formula for carbon tetrachloride?
Carbon tetrachloride, also known by many other names (such as tetrachloromethane, also recognised by the IUPAC, carbon tet in the cleaning industry, Halon-104 in firefighting, and Refrigerant-10 in HVACR) is an organic compound with the chemical formula CCl4.
Is water Polar?
Water is a Polar Covalent Molecule The unequal sharing of electrons between the atoms and the unsymmetrical shape of the molecule means that a water molecule has two poles – a positive charge on the hydrogen pole (side) and a negative charge on the oxygen pole (side).
Does CBR have a dipole?
And thus the C−Cl bond is POLAR. But molecular polarity is the result of the VECTOR sum of the individual bond dipoles. And thus when we sum geometrically in a vector fashion, the RESULTANT bond dipole is ZERO…………. And thus CCl4 and CBr4 are NON-POLAR molecules, whereas HCCl3 and CH2Cl2 are POLAR……..
What type of chemical bond is CU?
All bonds, and particularly Cu–S, are highly covalent in character.
What ion is no3?
Nitrate is a polyatomic ion with the chemical formula NO−3. Salts containing this ion are called nitrates. Nitrates are common components of fertilizers and explosives.
What is the name of of2?
PubChem CID24547Chemical SafetyLaboratory Chemical Safety Summary (LCSS) DatasheetMolecular FormulaF2O or OF2SynonymsOxygen difluoride Difluorine monoxide FLUORINE MONOXIDE Oxydifluoride Fluorine oxide More…Molecular Weight53.996
What is the correct name of Cl2O7?
PubChem CID123272StructureFind Similar StructuresMolecular FormulaCl2O7SynonymsChlorine heptoxide Dichlorine heptoxide Perchloric anhydride Chlorine oxide (Cl2O7) 12015-53-1 More…Molecular Weight182.90
What is the bond angle of ph3?
Name of the MoleculePhosphineMolecular FormulaPH3Hybridization TypeNilBond Angle93oGeometryTrigonal Pyramidal