What is the Carnot cycle used for

Charlotte Adams
Published May 11, 2026
Carnot cycle, in heat engines, ideal cyclical sequence of changes of pressures and temperatures of a fluid, such as a gas used in an engine, conceived early in the 19th century by the French engineer Sadi Carnot. It is used as a standard of performance of all heat engines operating between a high and a low temperature.
Why is the Carnot cycle useful?
The Carnot engine — or the Carnot cycle — is important because it describes a heat engine that uses reversible processes that can be handled theoretically. … If er > ec, then the net result would be the transfer of heat from a cold temperature to a high temperature. But this violates the Second Law.
What is Carnot cycle as used in heat engines?
A Carnot heat engine is a theoretical engine that operates on the Carnot cycle. … A heat engine acts by transferring energy from a warm region to a cool region of space and, in the process, converting some of that energy to mechanical work. The cycle may also be reversed.
Where do we use Carnot cycle?
The Carnot cycle is the absolute temperature of the hot reservoir. This definition of efficiency makes sense for a heat engine, since it is the fraction of the heat energy extracted from the hot reservoir and converted to mechanical work. A Rankine cycle is usually the practical approximation.Why is Carnot cycle not used in real applications?
Carnot cycle is not used in real engine because Carnot cycle contains four reversible process but in actually we can’t obtain reversible process . So we can’t use Carnot cycle in engines.
During which stages of the Carnot cycle is useful work extracted?
During which stages of the Carnot Cycle is useful work extracted? Stages 1 and 2 (isothermal expansion, and adiabatic expansion).
Why is Carnot cycle not used in steam power plant?
However; the Carnot cycle is not a suitable model for steam power cycle since: The turbine has to handle steam with low quality which will cause erosion and wear in turbine blades. … Thus, the Carnot cycle cannot be approximated in actual devices and is not a realistic model for vapor power cycles.
What is a Carnot cycle find the expression for the work done by a Carnot engine find its efficiency?
efficiency =WQH=1−TCTH. These temperatures are of course in degrees Kelvin, so for example the efficiency of a Carnot engine having a hot reservoir of boiling water and a cold reservoir ice cold water will be 1−(273/373)=0.27, just over a quarter of the heat energy is transformed into useful work.How is entropy lost?
5 Irreversibility, Entropy Changes, and “Lost Work” In general, reversible processes are accompanied by heat exchanges that occur at different temperatures. … in Equation (6.4)) is always greater than zero, so the only way to decrease the entropy of a system is through heat transfer.
What is Carnot cycle and its efficiency?P6.5, and the temperatures of the two reservoirs associated with the cycle are shown as TH and TC. The thermal efficiency of a Carnot cycle operating between these two reservoirs is η = 1−TC/TH. This value is significantly higher than that of the Otto cycle operating between the same reservoirs.
Article first time published onCan you design an engine of 100 efficiency?
It is impossible for heat engines to achieve 100% thermal efficiency () according to the Second law of thermodynamics. This is impossible because some waste heat is always produced produced in a heat engine, shown in Figure 1 by the term.
Why are Carnot engines not 100 efficient?
In order to achieve 100% efficiency (η=1), Q2 must be equal to 0 which means that all the heat form the source is converted to work. Hence negative temperature of absolute scale is impossible and we cannot reach absolute 0 temperature. …
What is the purpose of reheat cycle?
The purpose of a reheating cycle is to remove the moisture carried by the steam at the final stages of the expansion process. In this variation, two turbines work in series. The first accepts vapor from the boiler at high pressure.
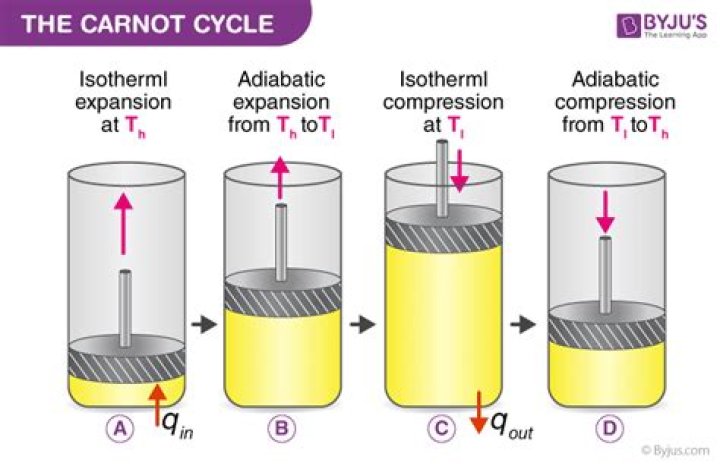
What are the four processes of the Carnot cycle?
The four stages in the Carnot cycle. (A) Stage 1: Isothermal expansion under heat input Q1, (B) Stage 2: Adiabatic expansion accompanied by a fall in temperature T1 to T2, (C) Stage 3: Isothermal compression, Q2 exhausted, (D) Stage 4: Adiabatic compression accompanied by an increase in temperature T2 to T1.
What are the two statements known as the Carnot principles?
Carnot principles can be expressed as the following two statements: 1. The efficiencies of all reversible heat engines operating between a constant temperature source (TH) and a constant temperature sink (TL) are the same. They only depend on the high temperature TH=const and the low temperature TL=const. 2.
When water freezes the entropy of the water?
When water freezes its entropy decreases. This does not violate the second law of thermodynamics. The second law does not say that entropy can never decrease anywhere. It just says that the total entropy of the universe can never decrease.
What is enthalpy in thermodynamics?
enthalpy, the sum of the internal energy and the product of the pressure and volume of a thermodynamic system. … In symbols, the enthalpy, H, equals the sum of the internal energy, E, and the product of the pressure, P, and volume, V, of the system: H = E + PV.
What is Exergy in thermodynamics?
Exergy (Ex) is defined as the amount of work (=entropy-free energy) a system can perform when it is brought into thermodynamic equilibrium with its environment, i.e. lAmax l=Ex (Jørgensen et al., 1999).
What is Carnot cycle in chemistry?
Definition of Carnot cycle : an ideal reversible closed thermodynamic cycle in which the working substance goes through the four successive operations of isothermal expansion to a desired point, adiabatic expansion to a desired point, isothermal compression, and adiabatic compression back to its initial state.
What is a Carnot engine explain briefly its construction draw Carnot cycle for a heat engine and obtain expression for efficiency of a Carnot engine?
Carnot engine is a theoretical thermodynamic cycle proposed by Leonard Carnot. It gives the estimate of the maximum possible efficiency that a heat engine during the conversion process of heat into work and conversely, working between two reservoirs, can possess.
Why is a perfect heat engine Impossible?
It is impossible to construct a perfect heat engine. … Since it absorbs heat, its entropy is increased. If it increases its entropy enough, it will ensure that the entire system has a net increase in entropy.
Is it possible to construct a heat engine that doesn t?
It is not possible to construct a heat engine which is free from thermal pollution.
What makes it impossible for a heat engine to work with 100%?
A heat engine is considered to be 100% efficient if only all the heat is converted into useful work or mechanical energy. Since heat engines cannot convert all the heat energy into mechanical energy, their efficiency can never be 100%.
Does Carnot engine exist?
as the name suggest it works on Carnot Cycle. this cycle was suggested by Nicolas Léonard Sadi Carnot in 1824. As it is a theoretical engine it has no real existence.
What is the principle of Carnot engine class 11?
Carnot Engine Principles The efficiency of an irreversible heat engine is always less than the efficiency of a reversible one operating between the same two reservoirs. The efficiencies of all reversible heat engines operating between the same two reservoirs are the same.
Can the efficiency of Carnot engine be 0 or 1?
Not only a Carnot engine, any energy converting machine or device cannot work with 100% efficiency for all obvious reasons . any engine can not be 100% efficient. so carnot engine’s efficiency can’t be 1 but less than 1 and that will be maximum for fixed temperature limits.
Why both reheating and regeneration is used together?
Explanation: When vaporization pressure is high, the reheating of steam is adopted. 2. Why both reheating and regeneration is used together? Explanation: Thus a modern steam power plant has both reheating and regeneration.
Which basic cycle is used for steam power plant?
The steam Rankine cycle is one of the highest utilized types of thermodynamic cycles for electricity production in the world [1]. The steam power stations run on Rankine cycles using steam as a working fluid. The simple ideal Rankine cycle is in fact the most basic vapor power cycle.
What is the function of regenerator?
Regenerators are compact heat exchangers in which heat is alternately stored and removed using a heat storage matrix. During the heating period, the hot gas passes through the regenerator and transfers heat to the matrix.