What is an enolate equivalent

William Cox
Published Apr 20, 2026
Imine anions and eneamines are synthetic equivalents of enolate anions.
Do esters form enols?
As we mentioned, esters can also form enolates, though in smaller concentrations than aldehydes or ketones. These enolates can then react with unreacted starting material in a condensation reaction that is exactly analogous to the aldol reaction.
What Cannot form an enolate?
q Since carbonyl compounds which do not have alpha hydrogens can not form an enolate, they cannot undergo the aldol reaction. Therefore the simplest aldehyde, methanal (formaldehyde) cannot undergo the aldol reaction.
What is the difference between an enol and enolate?
An enolate is the conjugated base of an enol. … The key difference between enols, enolates and enamines is that enols contain a hydroxyl group with an adjacent C=C double bond and enolates contain a negative charge on the oxygen atom of an enol whereas enamines contain an amine group adjacent to a C=C double bond.How do you make an enolate?
ENOLS CAN BE FORMED ONLY FROM CARBONYL COMPOUNDS WHICH HAVE ALPHA HYDROGENS. THEY CAN BE FORMED BY ACID OR BASE CATALYSIS, AND ONCE FORMED ARE HIGHLY REACTIVE TOWARD ELECTROPHILES, LIKE BROMINE.
Are Hemiacetals stable?
Only a few hemiacetals are stable Like their hydrates, the hemiacetals of most ketones (sometimes called hemiketals) are even less sta- ble than those of aldehydes.
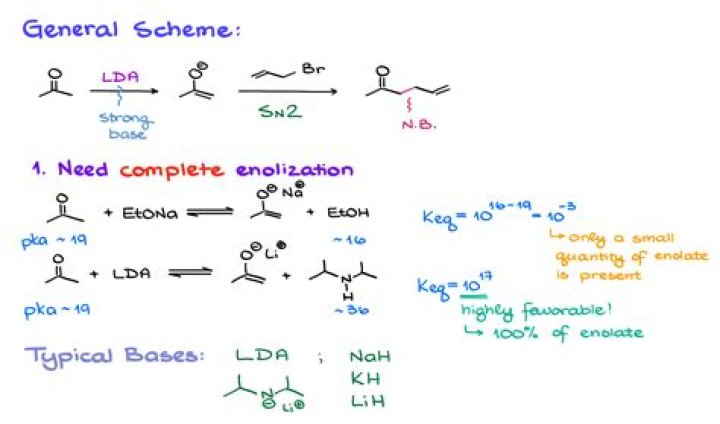
What does LDA and THF do?
Ether solvents like tetrahydrofuran (THF) are commonly used for enolate anion formation. With the exception of sodium hydride and sodium amide, most of these bases are soluble in THF. … Because of its solubility in THF, LDA is a widely used base for enolate anion formation.
What is enol and keto?
In organic chemistry, keto–enol tautomerism refers to a chemical equilibrium between a keto form (a ketone or an aldehyde) and an enol (an alcohol). The keto and enol forms are said to be tautomers of each other.Why is enol unstable?
Enols are unstable for a couple reasons. First, the free electrons on the oxygen can act as a base or nucleophile for chemical reactions. More commonly, one must consider that any time you have an enol, it can undergo tautomerization, a conversion to what is called the keto form.
Why is keto more stable than enol?In most keto-enol tautomerisms, the equilibrium lies by far toward the keto form, indicating that the keto form is usually much more stable than the enol form, which can be attributed to the feet that a carbon-oxygen double bond is significantly stronger than a carbon-carbon double bond.
Article first time published onCan aldehydes form Enolates?
The reaction involves an aldehyde enolate reacting with another molecule of the aldehyde. Remember enolates are good nucleophiles and carbonyl C are good electrophiles. Since the pKa of an aldehyde is close to that of NaOH, both enolate and aldehyde are present simultaneously.
Is enolate a Carbanion?
In carbanion: Delocalized ions. Enolate ions are derivatives of ketones and aldehydes (compounds containing a double bond between carbon and oxygen atoms), from which they can be generated by abstraction of a proton from the carbon atom that is located next to the carbon of the carbonyl group.
Are enolates strong nucleophiles?
“the conjugate base is always a better nucleophile”. I said enols were good nucleophiles. But their conjugate bases – enolates – are even better. … We call this an enolate.
What is the meaning of Enolizable?
Filters. (organic chemistry) That may be converted into an enol or enolate.
Can you Deprotonate an aldehyde?
If you attempt to deprotonate acetaldehyde, clearly the second option is better. It doesn’t exceed an octet for oxygen. The electrons are able to reasonably delocalize. The electronegativity of oxygen is greater than that of carbon, so oxygen is capably negatively charged.
How is an enolate ion formed?
An enolate ion is the anion formed when an alpha hydrogen in the molecule of an aldehyde or a ketone is removed as a hydrogen ion.
What is non Enolizable?
A non-enolizable aldehyde is an aldehyde whose molecule has no alpha hydrogens.
What is LDA used for chemistry?
Lithium diisopropylamide (commonly abbreviated LDA) is a chemical compound with the molecular formula [(CH3)2CH]2NLi. It is used as a strong base and has been widely utilized due to its good solubility in non-polar organic solvents and non-nucleophilic nature.
What is Hmpa in organic chemistry?
Infobox references. Hexamethylphosphoramide, often abbreviated HMPA, is a phosphoramide (an amide of phosphoric acid) with the formula [(CH3)2N]3PO. This colorless liquid is a useful reagent in organic synthesis.
What is the role of LDA in enolate formation?
Strong organic bases such as LDA (Lithium DiisopropylAmide) can be used to drive the ketone-enolate equilibrium completely to the enolate side. LDA is a strong base that is useful for this purpose. The steric bulk of its isopropyl groups makes LDA non- nucleophilic. Even so, it’s a strong base.
How do hemiacetals form?
The hemiacetal forms when an aldehyde reacts with an alcohol. This can occur with neutral reaction, which only involves the alcohol and the aldehyde, or an acid catalyzed reaction, which puts a hydrogen on the aldehyde oxygen to start out with and is much faster.
Are hemiacetals reducing sugars?
A hemiacetal form is thus a reducing sugar. In contrast, acetal forms (glycosides) are not reducing sugars, since with base present, the acetal linkage is stable and is not converted to the aldehyde or hemiacetal. The outcome is that in a reducing sugar the anomeric carbon is in an aldehyde or hemiacetal.
Can hemiacetals be oxidized?
Hemiacetal oxidation presents a possible alternative to acid-alcohol esterification as a biogenetic pathway for the formation of certain long chain esters.
What is meant by enol?
Definition of enol : an organic compound that contains a hydroxyl group bonded to a carbon atom having a double bond and that is usually characterized by the grouping C=C(OH)
Are Enols acidic?
In particular, α hydrogens are weakly acidic because the conjugate base, called an enolate, is stabilized though conjugation with the π orbitals of the carbonyl. … Of the two resonance structures of the enolate ion the one which places the negative charge on the oxygen is the most stable.
Why are Enolates more stable than Enols?
Answer: In most keto-enol tautomerisms, the equilibrium lies by far toward the keto form, indicating that the keto form is usually much more stable than the enol form, which can be attributed to the feet that a carbon-oxygen double bond is significantly stronger than a carbon-carbon double bond.
Why are ketones favored enol?
Which is More Stable, Keto Or Enol? Despite being a reversible reaction, the keto form is more stable and thus favored by equilibrium. This is because carbon double bound to oxygen is more stable than a carbon single bound to oxygen and single bound to hydrogen.
What does enol Tautomer mean?
Keto-enol tautomerism refers to a chemical equilibrium between the keto form (carbonyl structure containing α-hydrogen) and the enol form (a double bond adjacent to an alcohol, -C=C-OH) of a compound. The enol and keto forms are said to be tautomers of each other.
Why is keto form preferred?
In general , keto form is more stable than enol form due to greater bond energy of c=o. than c=c . But, in some cases enol form is more stable than keto . For example, if bulky phenyl groups are present at sp3 alpha carbon then enol is more stable than keto form due greater bond angle.
Why is enol form of acetoacetic ester more stable than it's keto form?
because of hydrogen bonding a 6 membered ring is formed in enol form ,which is more stable. hence it is more stable than keto form. Generally, whenever keto form are stable its because of greater C=O. bond energy than that if C=C.
Why does Tautomerism occur?
Mechanism of Tautomerization Either acid or base can catalyze proton transfer. Hence, tautomerization occurs by two different mechanisms. In the first step of acid-catalyzed tautomerization of the keto form, hydronium ion protonates the carbonyl oxygen atom. Then, water removes the α-hydrogen atom to give the enol.



