Does density increase temperature

Charlotte Adams
Published Apr 25, 2026
Density increase as the temperature decreases. … This is the reason why liquid water is more dense than solid water. The bonds in water break more slowly as temperature decreases and the structure tend to trap fewer extra water molecules. At low temperature, more of the water has the same lattice as ice.
What happens to density when temperature increases?
Density is directly proportional to pressure and indirectly proportional to temperature. … Conversely when temperature increases, with pressure constant, density decreases.
What is the relationship between the temperature and the density of a substance?
Temperature and density are inversely related. This means that when the temperature is increased, the density will decrease and vice-versa.
Does density increase or decrease as temperature increases explain?
With few exceptions, density decreases as temperature increases. Density is mass divided by volume. As you heat something up, its volume usually increases. The faster moving molecules are further apart.Why does density increase with temperature?
Cooling a substance causes molecules to slow down and get slightly closer together, occupying a smaller volume that results in an increase in density. Hot water is less dense and will float on room-temperature water. Cold water is more dense and will sink in room-temperature water.
What happens to density when the temperature decreases?
When temperatures increase, objects expand and become larger and therefore the density decreases. When temperatures decrease, objects condense and become smaller so density increases.
Why is density inversely proportional to temperature?
Density is inversely proportional to temperature because increasing temperature always causes the decompression of particles.
Does the density of a gas increase or decrease as the temperature increases and pressure is held constant explain?
So right from the start you can say that since the pressure of the gas is increased, the volume will decrease, which in turn will cause the density of the gas to increase, since now you have the same mass of gas in a smaller volume.Why does the density of gas changes with increase in temperature?
Answer: Since there are now more particles per cubic unit of length, the density increases. Also, when we increase the temperature of the gas molecules, we increase the energy of the molecules. As such, the Kinetic energy of the Molecules increase, which means the volume increases.
How does density vary with temperature quizlet?How does the density vary with temperature? The density of a substance generally decreases as it’s temp. increases. A weather balloon is inflated to a volume of 2.2 x 10^3 L with 37.4g of helium.
Article first time published onDoes the density of a solid change with temperature?
When a substance is heated its volume increases and so the density decreases. In solids, increase in volume is negligible and hence decrease in density too. In liquids and gases, as the temperature increases, volume increases and therefore density decreases considerably.
Why does density of liquid vary with temperature?
When the water is heated, it expands, increasing in volume. … The warmer the water, the more space it takes up, and the lower its density. When comparing two samples of water with the same salinity, or mass, the water sample with the higher temperature will have a greater volume, and it will therefore be less dense.
What is density how it varies with temperature?
As the temperature increases, volumes of most of the liquids also increases and when the volume increases density decreases. Similarly, when temperature decreases, the volume of most liquids decreases which increases the density.
What causes density to increase or decrease?
The density of a material varies with temperature and pressure. … Increasing the pressure on an object decreases the volume of the object and thus increases its density. Increasing the temperature of a substance (with a few exceptions) decreases its density by increasing its volume.
Does density affect melting point?
It is not a major factor in defining melting point. … A high density and high melting point require a low reactivity; equally, low density and low melting point require high reactivity. The melting point is similar to the heat of sublimation; the density provides a guide to the magnitude of the ionization energy.
Is density linear with temperature?
The relation between density and temperature is inversely proportional. …
Why does density change slightly with changes in temperature and pressure?
Density changes with temperature because volume changes with temperature. Density is mass divided by volume. As you heat something up, the volume usually increases because the faster moving molecules are further apart. Since volume is in the denominator, increasing the volume decreases the density.
How does increasing the density of a gas affect?
Increasing density of a gas affects its air pressure by increasing the air pressure.
How would the density of a gas be affected if the temperature decreased?
Thus, if the temperature of the gas is decreased, then its density will be increased.
Why does density increase when pressure increases?
Density is directly proportional to pressure and indirectly proportional to temperature. As pressure increases, with temperature constant, density increases.
How would the density of a gas change if the temperature of the gas was decreased?
Density of a gas can be termed as number of molecules per unit volume of the material. With increase in temperature, number of molecules the gas contains per unit volume of it decreases. So the density of the gas decreases with increase in temperature of the gas.
How does decreasing temperature affect air density?
As the temperature of air increases, its volume increases and its density decreases. As the temperature of air decreases, its volume decreases and its density increases. … As air is cooled, its density increases because its mass remains constant but its volume decreases.
What happens to the density of the air as it is heated?
2. How does the temperature of air affect air density? … So air, like most other substances, expands when heated and contracts when cooled. Because there is more space between the molecules, the air is less dense than the surrounding matter and the hot air floats upward.
What determines density quizlet?
Terms in this set (13) Density is the measure of how much mass is contained in a given volume of a substance. Density= mass/volume or d=m/v. Density is mass divided by volume. Density is expressed as a ratio of two different units.
Why is density of solid remains constant for minor changes in temperature?
The density of solids remain constant for minor changes in temperature because the particles of solids are closely packed with each other.
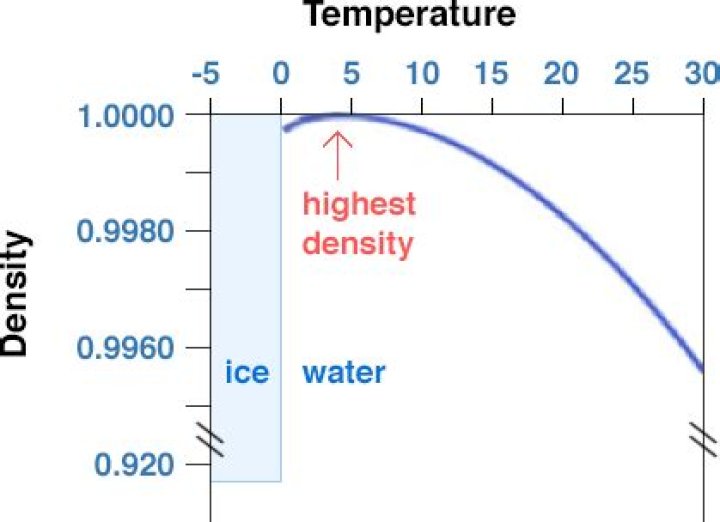
Why does the density of water decrease as temperature decreases?
A decrease in temperature caused the water molecules to lose energy and slow down, which results in water molecules that are closer together and a decrease in water volume. When water is heated, it expands, or increases in volume. When water increases in volume, it becomes less dense.
Why the density of liquid decreases with increase in temperature True or false?
In general, the liquids tend to expand when their temperature increases. For example, the same mass of boiling water occupies more volume at 100 degrees Celsius than at 20 degrees Celsius. Therefore, increasing temperature decreases density.