Does cyclobutane have ring strain

Andrew Vasquez
Published Apr 22, 2026
These destabilizing effects, angle strain and torsional strain are known together as ring strain. The smaller cycloalkanes, cyclopropane and cyclobutane, have particularly high ring strains because their bond angles deviate substantially from 109.5° and their hydrogens eclipse each other.
Which types of ring strain are present in cyclobutane?
Cyclobutane is a cyclic molecule with 4 Cs on it. It follows a square-like shape which is highly angle strained because C follows a tetrahedral geometry which has a bond angle around 109° and a square has around 90°. Torsional strain is also present in cyclobutane which is due to eclipsing of Hs on adjacent Cs.
Does cyclobutane have steric strain?
Cyclobutane is still not large enough that the molecule can reach around to cause crowding. Steric strain is very low. Cyclobutanes are a little more stable than cyclopropanes and are also a little more common in nature.
What is the angle strain of cyclobutane?
So, deviation of the bond angle (angle strain) in. cyclobutane =109∘28′−90∘/2.How is cyclobutane made?
Cyclobutane photodimers (CPD) are formed by photochemical reactions that result in the coupling of the C=C double bonds of pyrimidines. Thymine dimers (T-T dimers) formed in between two thymines are the most abundant of the CPDs.
Does cyclobutane have more torsional strain than cyclopentane?
Cyclopentane. Cyclopentanes are even more stable than cyclobutanes, and they are the second-most common cycloalkane ring in nature, after cyclohexanes. Planar cyclopentane has virtually no angle strain but an immense amount of torsional strain.
What accounts for the large amount of ring strain in cyclopropane and cyclobutane?
Both cyclopropane and cyclobutane have large ring strain due to a mixture of angle strain and torsional strain.
How do you calculate ring strain?
Ring strain is calculated as an energy difference between the molecule of interest and a reference species, plus/minus an additional strain-free energy term balancing the difference in the number of atoms and bonds in the two molecules.Which compound has highest ring strain?
Explanation: Cyclopropane is the compound with the highest ring strain. This is because the carbon atoms are arranged in the shape of a triangle thus forming C-C-C.
What increases ring strain?Ring Strain occurs because the carbons in cycloalkanes are sp3 hybridized, which means that they do not have the expected ideal bond angle of 109.5o ; this causes an increase in the potential energy because of the desire for the carbons to be at an ideal 109.5o.
Article first time published onIs cyclobutane a planar?
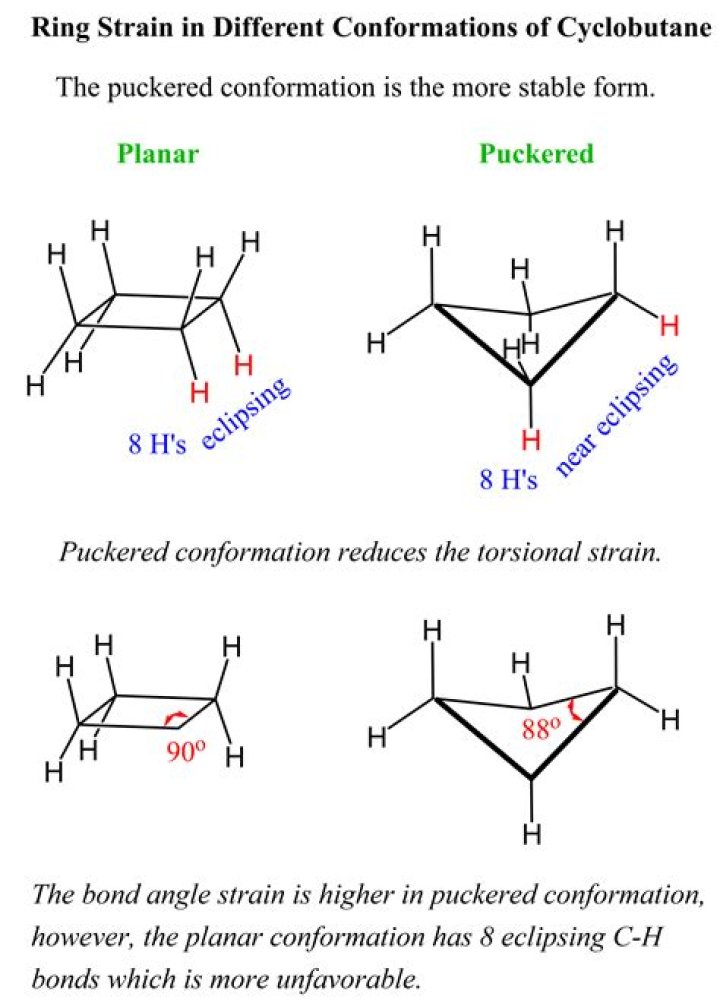
Cyclobutane is not planar. If it were, there would be eight pairs of eclipsed hydrogen atoms, which would account for 8 × 4.2 = 33.6 kJ mole− 1 of the total strain energy of cyclobutane.
Why cyclopropane is more reactive than cyclobutane?
In cyclobutane molecule the C-C-C bond angle is 90° while it is 60° in cycloprpane. This shows that the deviation from the tetrahedral bond angle 109° 28 in cyclobutane is less than in cyclopropane. In other words cyclopropane is under great strain compared with cyclobutane and is therefore more reactive.
Is cyclobutane flat?
Cyclobutane has considerable angle strain, but not as much as in cyclopropane. Unlike cyclopropane, which is flat, cyclobutane puckers to lessen somewhat (not eliminate, however) torsional strain. Puckering allows the bonds to remain only partially eclipsed.
Which of the Cycloalkanes has the greatest ring strain?
Ring strain is determined by the level of “strain” between the bonds of cycloalkanes. For example, propane has the highest ring strain of all cycloalkanes.
Is cyclobutane a liquid?
Cyclobutane is a cycloalkane and a member of cyclobutanes. Gas that condenses to a liquid at 55°F. Insoluble in water.
Is cyclobutane an alcohol?
Cyclohexanol is an alcohol that consists of cyclohexane bearing a single hydroxy substituent. The parent of the class of cyclohexanols. It has a role as a solvent. It is a secondary alcohol and a member of cyclohexanols.
Why are cyclopropane and cyclobutane rings very unstable compared to cyclopentane?
This bond angle causes cyclopropane and cyclobutane to be less stable than molecules such as cyclohexane and cyclopentane, which have a much lower ring strain because the bond angle between the carbons is much closer to 109.5o.
What is the lowest energy conformation of cyclobutane?
The lowest energy conformation of cyclobutane is known as the chair conformation. C) The lowest energy conformation is one in which the bond angles are slightly less than 90° even though this increases angle strain.
How many such interactions are present in cyclopropane?
Cyclopropane has six pairs of hydrogen–hydrogen eclipsed interactions. We recall that a hydrogen–hydrogen eclipsing interaction in ethane is 4.2 kJ mole− 1. Although the bond angles are not the same in cyclopropane as in ethane, we can estimate that the total torsional strain energy is 6 × 4.2 = 25.2 kJ mole− 1.
What is ring strain in organic chemistry?
In organic chemistry, ring strain is a type of instability that exists when bonds in a molecule form angles that are abnormal. Strain is most commonly discussed for small rings such as cyclopropanes and cyclobutanes, whose internal angles are substantially smaller than the idealized value of approximately 109°.
Why is cyclopentane more stable than cyclobutane?
The next in the series is cyclobutane, which is also highly strained. Cyclopentane is slightly more stable because it can relieve some of the bond strain by buckling the ring and pushing one carbon out of the plane.
Which is more acidic cyclopropane or cyclobutane?
The smaller the internal angle is as observed in smaller cycloalkanes, the greater S character in C-H bonds hence greater acidic character. So, order of acidic character is cyclopropane > cyclobutane > cyclopentane.
What is baeyer's strain?
strain theory, in chemistry, a proposal made in 1885 by the German chemist Adolf von Baeyer that the stability of carbocyclic compounds (i.e., those of which the molecular structure includes one or more rings of carbon atoms) depends on the amount by which the angles between the chemical bonds deviate from the value ( …
Why is cyclohexane free of ring strain?
The flexibility of cyclohexane allows for a conformation which is almost free of ring strain. … In this conformation, the carbon-carbon ring bonds are able to assume bonding angles of ~111o which is very near the optimal tetrahedral 109.5o so angle strain has been eliminated.
Is cyclobutane heterocyclic?
Four-membered heterocycles are the heterocyclic analogs of cyclobutane and are considered to be derived by replacing a -CH2 (methylene group) by a heteroatom (NH, O or S).
How many atoms of carbon are there in cyclobutane?
Carbon groups that are attached to the ring are named as alkyl groups. Thus, the name “cyclobutane” indicates a ring of four carbon atoms, all linked by single bonds.
Is Cyclobutane more reactive than butane?
In order to relieve this tremendous angle strain cyclopropanes engage in high reactivity than straight–chain propane. This effect is less pronounced in cyclobutane, which has a bond angle of 90°, Cyclobutane forms a square molecule, which is less reactive than cyclopropane, but is more reactive than butane.
Is cyclopropane more stable than Cyclobutane?
Because Cyclopropane has less bond strength so it is unstable.
Why is cyclopropane not stable?
The bonding in cyclopropane is extremely strained. … Instead of aligning colinearly or parallel, the orbital alignment here is bent, creating an in-between stability and strength—forming bonds that are weaker than σ bonds, but stronger than π bonds.
What is torsional strain?
Torsional strain is the resistance to bond twisting. In cyclic molecules, it is also called Pitzer strain. Torsional strain occurs when atoms separated by three bonds are placed in an eclipsed conformation instead of the more stable staggered conformation.